Introduction: Unveiling the Versatility of Acetic Acid
Acetic acid, scientifically known as ethanoic acid, and also referred to as ethylic acid or methane carboxylic acid, stands as a fundamental organic compound with an expansive footprint across numerous industrial sectors and even biological systems. This colorless liquid, characterized by its distinctive pungent odor, is one of the most important carboxylic acids, playing a critical role in a myriad of applications ranging from large-scale chemical synthesis to intricate biological processes. Its significance is underscored by its diverse properties and the wide array of derivatives it forms, making it an indispensable component in modern manufacturing and development.
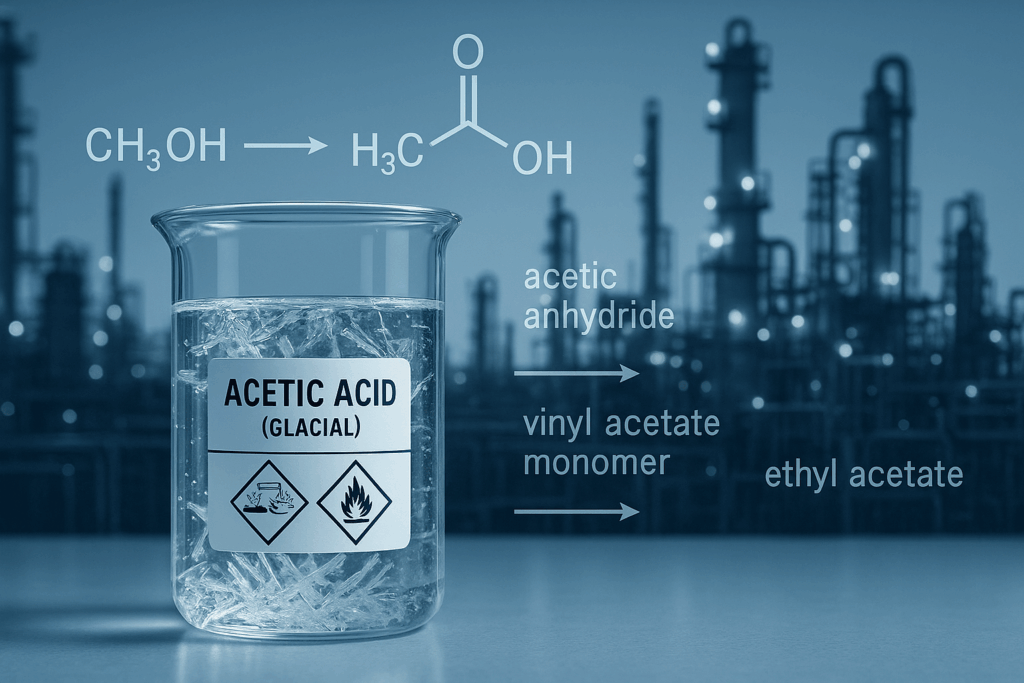
Historically, acetic acid has been recognized primarily in its dilute form as vinegar, a byproduct of fermentation. However, its industrial importance far transcends this common household use. In its purest, water-free (anhydrous) form, it is known as glacial acetic acid, a term derived from its ice-like appearance when frozen at temperatures below 16.6°C (61.9°F). While highly corrosive and demanding careful handling due to its potential to cause severe skin burns, permanent eye damage, and metal corrosion, its concentrated form unlocks a vast spectrum of industrial possibilities. The widespread production of acetic acid, predominantly through the carbonylation of methanol (accounting for approximately 75% of industrial output) and bacterial fermentation, highlights its pivotal position in the global chemical landscape. This blog post delves into the multifaceted world of acetic acid, exploring its key properties, diverse applications, and its overarching importance in driving various industries forward.
Key Properties and Characteristics
Understanding the properties of acetic acid is crucial to appreciating its extensive utility. With a chemical formula of CH₃COOH, a molecular formula of C₂H₄O₂, and a molar mass of 60.05 g/mol, acetic acid possesses a unique set of physical and chemical attributes that make it highly reactive and versatile.
- Physical State and Appearance: At room temperature, pure acetic acid is a clear, colorless liquid. Its anhydrous form, glacial acetic acid, is characterized by its ability to freeze into a solid resembling ice, hence its name. It is completely miscible with water, ethanol, and ether, indicating its excellent solvent capabilities.
- Acidity: As a carboxylic acid, acetic acid is a weak acid, meaning it does not fully dissociate in water. However, it is acidic enough to react with bases, alcohols, and other organic compounds, forming salts (acetates), esters, and other derivatives.
- Solvent Properties: Acetic acid is an excellent polar protic solvent. This property makes it widely used as a solvent for recrystallization processes, enabling the purification of various organic compounds. It is also a preferred solvent for reactions involving carbocations, such as Friedel-Crafts alkylation, where its protic nature facilitates the reaction.
- Corrosive Nature: Concentrated acetic acid is highly corrosive to metals and tissues. Direct contact with skin can lead to tissue damage, and inhalation of high concentrations of its vapors can cause irritation to the eyes, nose, and throat. Prolonged occupational exposure has been linked to conditions such as conjunctivitis, bronchitis, pharyngitis, and erosion of exposed teeth, underscoring the necessity of stringent safety protocols in its handling.
- Antifungal and Antibacterial Properties: Beyond its chemical reactivity, acetic acid exhibits significant antifungal and antibacterial properties. These biological attributes contribute to its use in medical and agricultural applications, where it can act as a bacteriostatic agent or an active ingredient in certain treatments.
- Industrial Specifications: Industrial-grade acetic acid, such as GNFC acetic acid, typically boasts a high purity of 99.85% minimum by weight. Other specifications include a water content of 0.15% maximum, a color of 10 APHA maximum, and minimal impurities like formic acid (0.05% max), acetaldehyde (0.05% max), and heavy metals (less than 2 ppm as Pb). Its freezing point is around 16.4°C, and its specific gravity is approximately 1.049 at 25°C. It is identified by CAS No. 64-19-7 and UN No. 2789.
Diverse Applications Across Industries
The broad spectrum of acetic acid’s properties translates into an equally wide array of applications across numerous industrial and commercial sectors. Its versatility makes it a cornerstone ingredient in the production of countless essential goods and processes.
Chemical Synthesis and Derivatives
Acetic acid is a critical building block in the synthesis of a multitude of important chemicals. Its derivatives are often more stable or possess specific properties required for various end uses.
- Acetic Anhydride: A key derivative, acetic anhydride, is primarily used in the production of cellulose acetate, a material vital for photographic films, textiles (e.g., acetate rayon), and cigarette filters.
- Vinyl Acetate Monomer (VAM): VAM is another major derivative, serving as a monomer for polyvinyl acetate (PVA) and vinyl acetate copolymers. These polymers find extensive use in adhesives, paints, coatings, and textiles.
- Acetic Esters: Various acetic esters, such as ethyl acetate, butyl acetate, and propyl acetate, are widely used as solvents in lacquers, paints, inks, and adhesives due to their excellent solvency and favorable evaporation rates.
- Chloracetic Acid: This derivative is important in the synthesis of pharmaceuticals, dyes, and herbicides.
- Plastics and Rubber: Acetic acid and its derivatives are integral to the manufacturing processes of various plastics and rubber products, contributing to their properties and functionalities.
- Dyes and Insecticides: It is a vital component in the synthesis of numerous dyes used in textile and other industries, and also plays a role in the production of certain insecticides.
- Photographic Chemicals: Historically, acetic acid was a critical component in photographic developing solutions, particularly as a stop bath to halt the development process.
Pharmaceuticals and Healthcare
The biological properties of acetic acid, especially its antifungal and antibacterial nature, lend themselves to several applications in the pharmaceutical and healthcare industries.
- Ear Canal Infections: Diluted acetic acid solutions are commonly used as eardrops or in ear wicks to treat outer ear canal infections, helping to reduce swelling and combat bacterial or fungal growth.
- Bladder Irrigation: It is used as a liquid to flush the bladder in individuals with urinary catheters, aiming to prevent blockages and infections.
- pH Adjustment: Acetic acid can be used to adjust the pH in certain medical applications, including maintaining vaginal pH balance.
- Cervical Cancer Screening: In a significant application, a diluted acetic acid solution is applied to the cervix during screening for cervical cancer. Abnormal cells, if present, will temporarily turn white, aiding in early detection.
- Bacteriostatic Agent: Its ability to inhibit the growth of bacteria makes it a valuable bacteriostatic agent in various medical solutions.
Agrochemicals and Agriculture
Acetic acid and its formulations are increasingly recognized in the agricultural sector for their efficacy in weed control and veterinary medicine.
- Herbicides: New acetic acid-based herbicides are being developed and utilized for broad-spectrum control of turfgrass, woody plants, and broadleaf weeds in non-crop areas and hard surfaces. These environmentally friendlier alternatives are gaining traction in landscape renovation situations.
- Veterinary Medicine: It also finds applications in veterinary medicine, although specific uses vary.
- Plant Tissue Desiccation: When applied to plants, acetic acid, commonly known as vinegar in this context, affects cell membranes, leading to rapid breakdown and desiccation of foliage tissue on contact.
Textiles and Dyes
The textile industry heavily relies on acetic acid for various processes related to dyeing and fabric treatment.
- Dyeing Agent: It is widely used in garment dyeing to regulate the pH of dye baths, which is crucial for achieving consistent color absorption and preventing dye degradation.
- Color Fixer: Textile companies utilize acetic acid as a color fixer, improving the colorfastness of dyed fabrics and ensuring the longevity of hues.
- Nylon Production: Acetic acid plays a role in the manufacturing process of nylon, a synthetic fiber known for its strength and durability.
- Specialized Dyeing: In printing and dyeing, acetic acid functions as an alkali-resistant agent for azoic dyes, an auxiliary for disperse dyes on polyester, and is used in the dyeing of mulberry silk with weak acid dyes, showcasing its specialized roles in achieving desired textile finishes.
Manufacturing and Industrial Processes
Beyond chemical synthesis, acetic acid finds a myriad of applications in various manufacturing and industrial settings.
- Cleaning Agent: It serves as a component of cleaning agents, particularly in the manufacturing of semiconductors, where precision cleaning is paramount.
- Lacquers: Acetic acid is used in the production of lacquers, which are clear or colored coatings used to protect and enhance surfaces.
- Synthetic Fibers, Rubber, and Plastics: It is an integral part of the manufacturing processes for synthetic fibers, rubber, and various plastic materials, contributing to their formulation and properties.
- Concrete Additive: During winter months, acetic acid can be used as an additive to concrete mixes to prevent premature hardening, allowing for more workable time.
- Wastewater Treatment: Acetic acid is increasingly employed in wastewater treatment processes, aiding in environmental protection by facilitating the removal of pollutants, reflecting a growing international trend towards sustainable industrial practices.
- Petroleum Production: It finds use in certain aspects of petroleum production, though specific details can vary depending on the particular process.
Biological Significance and Metabolism
While often discussed in an industrial context, acetic acid and its salts, known as acetates, are ubiquitous in biological systems and play a vital role in metabolism. Acetates are common components in both animal and plant tissues, primarily formed during the metabolism of food substances. Most tissues readily metabolize acetate, often leading to the production of ketones as intermediate products. The body strategically utilizes acetate as a foundational building block for synthesizing a wide array of crucial biological molecules, including:
- Phospholipids: Essential components of cell membranes.
- Neutral Lipids: Important for energy storage.
- Steroids and Sterols: Including cholesterol, vital for hormone production and cell structure.
- Saturated and Unsaturated Fatty Acids: Fundamental for energy, cell structure, and various physiological functions.
This extensive metabolic utilization of acetate across various human and animal tissue preparations underscores its fundamental importance in biological processes, bridging the gap between its industrial utility and its intrinsic role in life itself.
Conclusion: Acetic Acid – An Unsung Hero of Modern Life
From its humble origins as a byproduct of fermentation to its current status as a powerhouse chemical, acetic acid has proven its enduring value. Its unique blend of properties – its acidity, solvent capabilities, corrosive nature (when concentrated), and even its biological significance – makes it a compound of extraordinary versatility. Whether it’s driving the production of essential plastics and textiles, safeguarding health in pharmaceutical applications, aiding sustainable agriculture through innovative herbicides, or playing a fundamental role in our own metabolism, acetic acid is an unsung hero that underpins countless aspects of modern life.
As industries continue to evolve and demand more efficient, sustainable, and specialized chemical solutions, the importance of acetic acid is only set to grow. Its adaptability ensures its continued relevance in existing applications while paving the way for new discoveries and innovations. Companies like Prakash Chemicals Agencies Pvt. Ltd., by providing quality solutions in the realm of acetic acid, play a crucial role in ensuring its safe and effective deployment across diverse industrial landscapes. The story of acetic acid is a testament to the power of fundamental chemistry to shape our world, making it a truly indispensable compound for present and future advancements.
